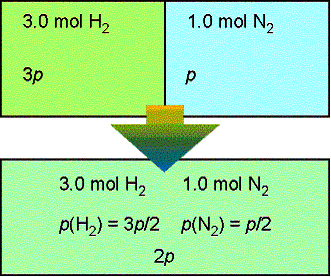
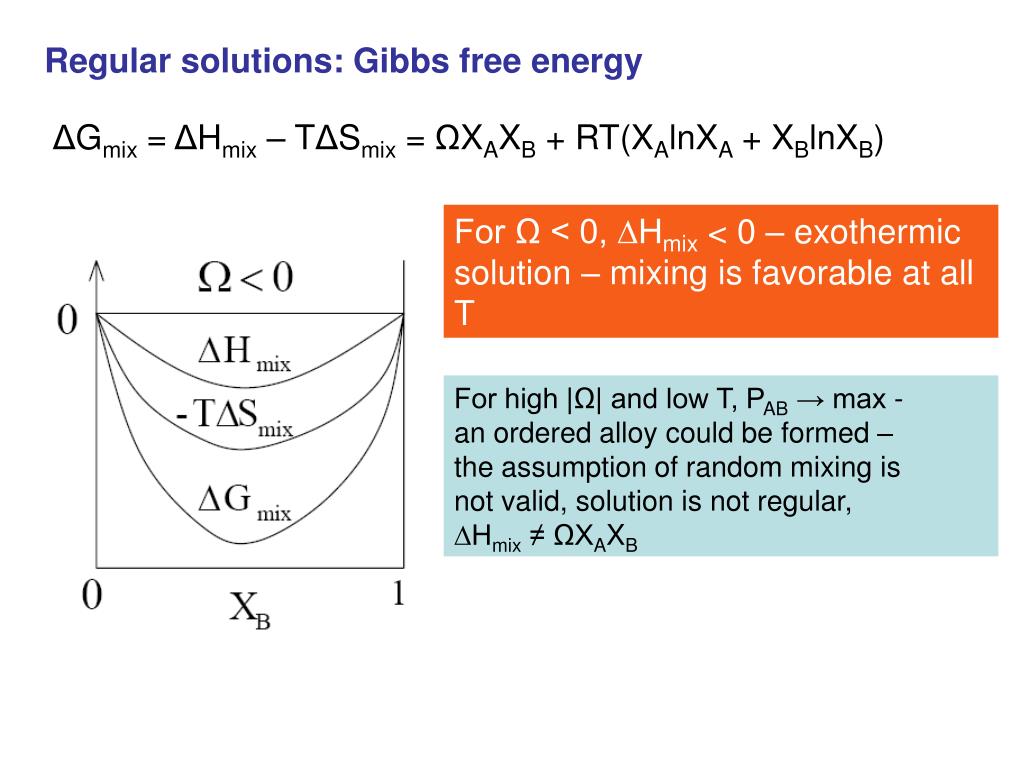
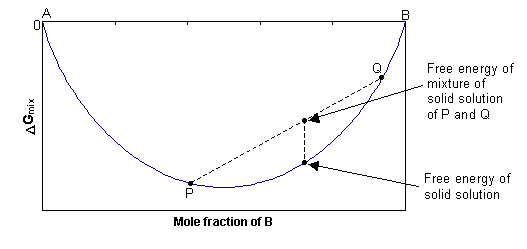
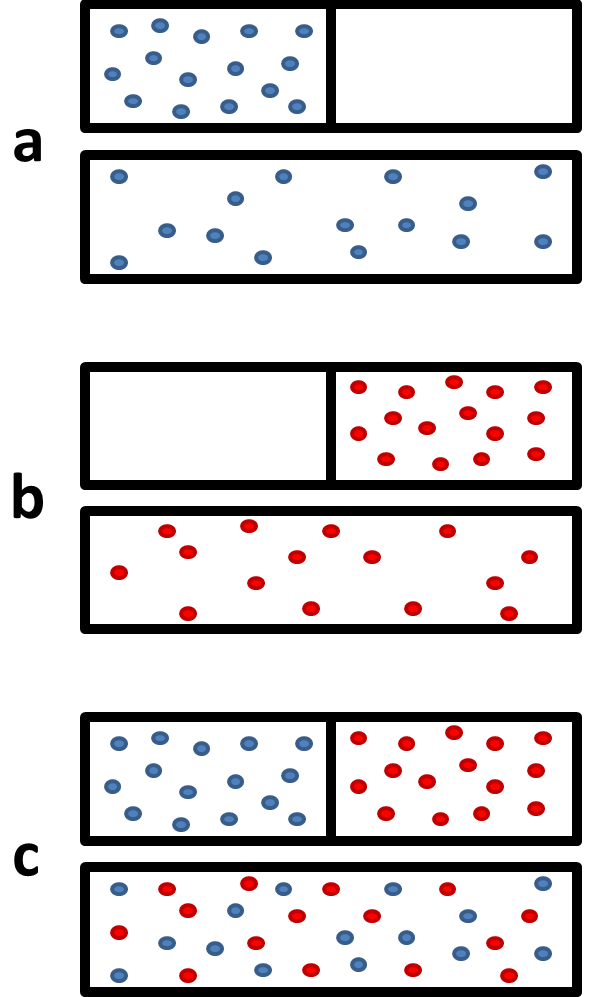
![Lecture 18Multicomponent Phase Equilibrium1 Theories of Solution The Gibbs energy of mixing is given by: And the chemical potential is: For ideal gases, - [PPT Powerpoint] Lecture 18Multicomponent Phase Equilibrium1 Theories of Solution The Gibbs energy of mixing is given by: And the chemical potential is: For ideal gases, - [PPT Powerpoint]](https://static.cupdf.com/img/365x274/reader020/slide/20190820/56649d425503460f94a1e10b/document-0.png)
Lecture 18Multicomponent Phase Equilibrium1 Theories of Solution The Gibbs energy of mixing is given by: And the chemical potential is: For ideal gases, - [PPT Powerpoint]
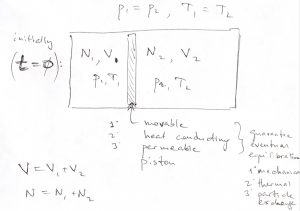
Gibbs Free Energy, Entropy of Mixing, Enthalpy, Chemical Potential, Gibbs-Duhem – Basic Notions of Thermodynamics and Quantum Mechanics for Natural Sciences

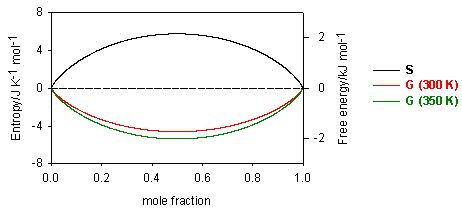


![PDF] On the prediction of Gibbs free energy of mixing of binary liquid alloys | Semantic Scholar PDF] On the prediction of Gibbs free energy of mixing of binary liquid alloys | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/81e2e777b45d3897628323bfc42a4f91db61d2f9/5-Table1-1.png)

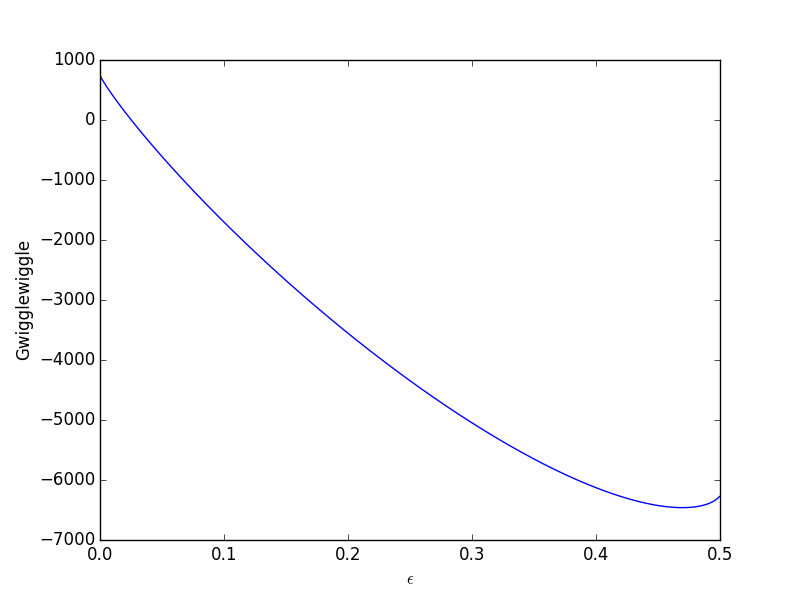
![The Gibbs energy of mixing, Delta g_mix [J/mol], in a | Chegg.com The Gibbs energy of mixing, Delta g_mix [J/mol], in a | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F471%2F47141c8f-8f8e-4905-9865-4b33d23f972b%2FphpR2OsNz.png)